|
The standard condition is to have a pH of 4 in the anode half cell but sometimes during nonstandard states, the pH may be higher or lower changing the voltage.This means that the reduction of chloride = 1.31V not 1.36V Express your answer in coulombs to three significant. 2 3 In the present version of the SI it is equal to the electric charge delivered by a 1 ampere constant current in 1 second and to 5 × 1027 801 088 317 elementary charges, e, (about 6.241 509 × 1018 e ). What is the charge q1 on the particle Ignore the effects of gravity. The coulomb (symbol: C) is the unit of electric charge in the International System of Units (SI). Concentration of chloride ion = 5.5M not the unit activity of 1M. Study with Quizlet and memorize flashcards containing terms like Suppose that a charged particle of diameter 1.00 micrometer moves with constant speed in an electric field of magnitude 1.00×105 newtons per coulomb while acted upon by a drag force of 7.25×1011 newtons.The reactants may be in nonstandard conditions which means that the voltage for the half cells may be less or more than the standard condition amount.There might be more than one electrode reaction that occurs meaning that there may be more than one half-reaction leaving two or more possibilities for the cell reaction.H 2 (g) requires a 1.5 V overpotential, while Pt (s) requires 0 V overpotential This case happens more frequently with gases. An overpotential or voltage excess is sometimes needed to overcome interactions at the electrode surface.There are four primary factors that determine whether or not electrolysis will take place even if the external voltage exceeds the calculated amount: If an aqueous solution of sodium chloride were used in the above system, hydrogen would undergo reduction instead of sodium, because it is a stronger oxidizing agent that sodium. The substance that is the strongest oxidizing agent will be reduced. The substance that is the strongest reducing agent (the substance with the highest standard cell potential value in the table) will undergo oxidation. The conditions under which the electrolyte cell operates are very important.In this episode, Shini talks about electrostatic forces, electrical charge, Coulombs. Anode is now positive charged and the cathode has a negative charged. Some examples of anions (ions with a negative charge) include: Chloride. Note that the site of oxidation is still the anode and the site of reduction is still the cathode, but the charge on these two electrodes are reversed. 
And whether such a capacitor could give you a lethal shock will again depend on the capacitance.+ 2e^- \nonumber \] Such a device can easily be charged with a Coulomb - although that isn't a net charge (one plate will be positive, the other negative). Supercapacitors can be created in which two conductors are brought in very close proximity, while having a dielectric layer in between that produces a very high capacitance in a small package. That's a very large voltage - if you could even maintain that potential (not in ordinary atmosphere), touching it would kill you. A sphere with a 1 m radius, with a 1 C charge on it, would have a potential of about 1 GV. Calculate the magnitude of the electrostatic force between the particles. So what is the size of a sphere with a capacitance of 1 F? Capacitance of a sphere is $4\pi\epsilon_0 r$, so you would need a radius of about $9\cdot 10^9 $ m - quite a bit bigger than the Earth. 0 c m distant from a second particle of charge 1. 
But if the voltage is high, it will easily overcome the resistance of your skin and give you an almighty jolt - possibly enough to kill you. If you have a charged object with a low potential, the flow of current through your body will be slow - and you will survive. An electrochemical unit of charge, the faraday, is useful in describing electrolysis reactions, such as in metallic. From the definition of the ampere, the electron itself has a negative charge of 1.602176634 × 10 19 coulomb. 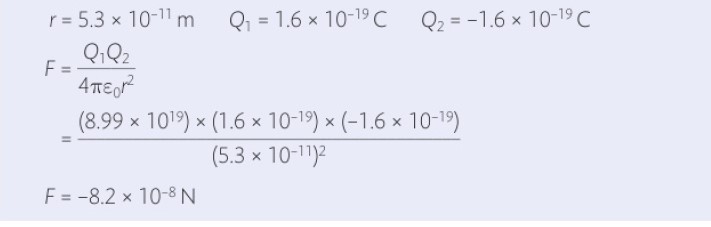
Avogadro's number is $6.02\cdot 10^$ - so for the same amount of charge, a larger capacitor will have less stored energy.Īnd that is the hint to the "will it kill me" part of the question: you are not killed by charge, but by current flowing. One coulomb consists of 6.24 × 10 18 natural units of electric charge, such as individual electrons or protons.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
